February 16, 2006
Cell Dynamics Take the Stage at Young Investigators’ Workshop
A worldwide effort to solve the ALS puzzle was captured by presentations and fruitful discussions at the Young Investigators’ Workshop January 23 through 25, 2006 in St. Petersburg, Florida, sponsored by The ALS Association (ALSA), graciously hosted by the ALSA Florida Chapter, and generously funded by The Linden Foundation in memory of Suzanne V.A. Kelsey. Investigators from many different countries who are just beginning to grapple with the disease used the opportunity to plan innovative approaches.
“It was an excellent opportunity for exchange of ideas and potential collaborations,” said Lucie Bruijn, Ph.D., ALSA science director and vice president.
The workshop was also an opportunity to present the The Milton Safenowitz Post-Doctoral Fellowship for ALS Research to two young investigators, Eran Perlson, Ph.D. and Agnès Lukaszewicz, Ph.D. ALSA holds these workshops every other year to foster an exchange of new ideas and promote collaboration. The conferences also encourage those new to the field to stay focused on the challenges posed by ALS (amyotrophic lateral sclerosis, also called Lou Gehrig’s disease).
Investigations into causes and possible treatment of ALS are converging on the dynamics within motor neurons and the other cells affected in the disease. A burden of damaged protein may somehow interrupt the flow of vital components, and one cellular victim could be the mitochondria that power metabolic processes. Also of note were findings on the potential role of the muscle in the disease that might turn into a therapeutic route. All represent avenues toward potential therapeutics, which are important to explore further. The workshop was a means to think clearly about potential approaches and chart future course.
For details, please click here.
-----
Report from the Young Investigators Workshop, St. Petersburg, Florida, January, 2006
The presentations at the 2006 ALSA Young Investigators Workshop spanned the range of suspect processes underlying ALS and ways to target potential therapies. The workshop began with an evening social, at which Kenneth Fischbeck, M.D., co-chair of the conference and chief of Neurogenetics at the National Institute of Neurological Disorders and Stroke (NINDS), gave an up-to-date view of ALS research. Some 40 different diseases affect motor neurons, the cells that die in ALS. Each mutation, even in different diseases, gives insight as an “experiment of nature” into the question of how motor neurons are vulnerable in ALS.
Strategies being pursued in ALS research include finding responsible genetic defects, so a therapy could be created to correct or compensate. Mitochondrial damage is increasingly documented in ALS. Evidence also suggests that too much glutamate may be bad for the cells; this is the basis for the action of riluzole in ALS.
Loss of supportive molecules that normally work to keep neurons healthy is another part of the picture of ALS, as is aggregation of protein. Strategies to address these include giving myotrophin (insulin-like growth factor or IGF-1) and other so called trophic factors in a way that allows them to reach the suffering cells and screening for compounds that would stop aggregation. Loss of the motor neurons may be addressed in the future by strategies to replace them, such as stem cell therapy.
At the workshop the following morning, a patient who graciously shared his symptoms and family history lent immediacy to the gathering. Kevin Boylan, M.D., of the recently certified ALSA center at the Mayo Clinic, Jacksonville, Florida, gave an overview of the clinical picture of ALS that allowed researchers from different areas to get on the same page in understanding the manifestations of the disease. This helped to set the stage for the reports by investigators that included mutant protein and the damage it might do in ALS, types of cells involved, cell dynamics, new targets for therapeutic approaches, and stem cell strategies.
Mutant Protein, Mitochondria, and Cell Players in ALS: (Mitochondria; SOD1 and ALS )
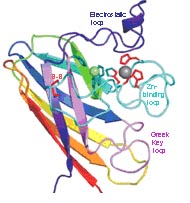 |
| Schematic of the SOD1 protein, Peter A. Doucette, Ph.D., and Joan Valentine, Ph.D. Reprinted, with permission, from the Annual Review of Biochemistry, Volume 74 ©2005 by Annual Reviews www.annualreviews.org. |
The mutations in a protein linked to some forms of ALS may impair the cell’s power plants, called the mitochondria, an idea that is gaining ground as a feature in the disease. Ashutosh Tiwari, Ph.D., of the University of Massachusetts Medical School, provided a plausible explanation of how more than 100 different mutations to the protein, copper-zinc superoxide dismutase (SOD1), can all cause an inherited form of ALS. He showed that the mutant proteins share a vulnerability to partial unfolding under conditions present in living cells. The normal enzyme is highly resistant to these stresses. Partially unfolded SOD1 cannot bind its metal ions properly and may interact with other cellular constituents to produce the damage seen in ALS. The toxic result may be to hamper the ability of mitochondria to power the cell, shown in findings reported in a yeast experiment by Michael Gunther, Ph.D. at West Virginia University. The yeast can yield abundant mitochondria for study that closely resemble mitochondria in people.
Scott Oakes, M.D., at the University of California San Francisco, is trying to determine how misfolded proteins such as mutant SOD1 lead to neuronal death through a genetically controlled form of "cell suicide" termed apoptosis. He has been able to detect the initial death signal that cells relay in response to prolonged or excessive protein misfolding. He is now attempting to biochemically purify and identify this early apoptotic signal in the hopes that blocking it would preserve neuronal viability in ALS patients.
Patricia Cassina, M.D., Ph.D. at the Universidad de la Republica in Montevido, Uruguay finds that mitochondria in the support cells called astrocytes are not able to process oxygen properly if obtained from rodents with mutant SOD1. Cassina has uncovered an interaction between astrocytes and motor neurons. In cell cultures, if the astrocytes are normal, then motor neurons extend their fibers. But astrocytes with SOD1 mutation do not support survival of motor neurons in the lab culture. Severine Boillee, Ph.D., working with
Transport of Materials within the Nerves
It is possible that a potentially toxic outcome of mutant SOD1 could be to change the movement of mitochondria along the long fiber connecting a motor neuron with its target muscle. Mitochondria are actively carried through the nerve fiber to arrive and work at the junction with muscle and also back to the cell body for metabolic tasks there. Andrew Grierson, Ph.D. at the University of Sheffield, U.K. has shown that in cultured rat nerve cells, mutated versions of SOD1 affect the distribution of mitochondria along the nerve fiber, called the axon. Motor proteins that move cellular materials along the axon of nerve cells usually move in only one direction. Kinesins move materials toward the growth cone as the axon reaches for its target cell. Dynein moves back toward the cell body (retrograde). In cells in culture, each of three different mutations to SOD1 gave a net loss of mitochondria at the nerve endings. Could the reduced energy available to the neuromuscular junction tilt the system toward degeneration?
Gerardo Morfini, Ph.D. also finds changes in axonal transport with SOD1 mutation using materials from squid nerve as an experimental setting to allow a microscopic view of transport in action.
 |
| The squid Loligo pealeii. Credit: Gerardo Morfini, Ph.D. and Scott Brady |
Damage to either kinesins or to dynein will produce problems in axon transport. The large axon in squid can allow researchers to see cell components move. The material within the axon can be removed and placed on a microscope slide, and the processes involved in transport are still intact and operating. Mutated protein can be directly introduced into this material. Preliminary data suggests that kinesins may play a role in SOD1-mediated disease and provide a potential target for therapeutic intervention.
Potential Environmental Toxin to Be Explored
Steven Stellman, Ph.D., M.P.H. of Columbia University presented the possible role of toxins in ALS, a topic that has repeatedly produced tantalizing but inconclusive findings for ALS. Risk factors for ALS include residence in Guam, where the decline in incidence suggests an environmental factor. Other risk factors include work with electrical equipment, including welding, and agricultural chemicals. Participation in the military is a recognized risk for ALS as is cigarette smoking.
Vietnam veterans are not yet included in the documentation for elevated risk with military service. What about the herbicides that were used during that war? Stellman will look at records of troop deployment and areas sprayed to investigate any link with incidence of ALS.
New Suggestions for Therapeutic Strategies
Chien-Ping Ko, Ph.D. of the University of Southern California reported on his provocative studies on the role of muscle in ALS, showing that a graft of muscle tissue from a normal rodent will survive and work properly, with appropriate nerve supply, in a SOD1 mutant. He can make muscle larger in the animals in another way by stopping the production of protein that normally keeps the size of muscle in check. Mutant SOD1 animals missing this so-called myostatin protein have larger muscles. While this gives the sick animals more strength, they still die at the same time as is typical for SOD1 mutants. Nevertheless, treating muscle might be a useful approach toward a better quality of life in ALS.
The new information on mitochondrial behavior and muscle’s role dovetails with earlier findings that at least some ALS patients appear to have increased muscle metabolism, according to workshop participant Luc Dupuis, Ph.D. of INSERM in Strasbourg, France. His team led by Jean-Philippe Loeffler, Ph.D. will see if there might be a way to modulate this metabolic state of the muscle.
Still another intriguing finding with therapeutic potential comes from the lab of Svitlana Garbuzova-Davis, Ph.D. at the University of South Florida who showed that infusion of mononuclear cells from human umbilical cord blood could modestly prolong the lifespan and delay onset of disease in SOD1 mutant mice. The effect due to the cell infusion may be a result of immune system modulation, in addition to anti-inflammatory effects.
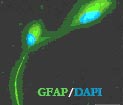 |
| Cells from human umbilical cord blood display markers that show they are neural cells. Image: Svitlana Garbuzova-Davis, Ph.D. |
Neither the muscle or cord blood strategies are ready for human trials. Far too little is understood about the cord blood action. Muscle grafts, easily accomplished in the inbred mice, would require massive immune suppression to even have a chance for safety in patients. Both do point investigators toward new routes to explore for eventual therapeutics.
Participant David Borchelt, Ph.D. of the University of Florida emphasized that a wider variety of models of the disease must be tested with candidate therapies. Many researchers use the mouse with the G93A mutation in SOD1. But many more mutations are available in mice, and many other model systems can recreate aspects of ALS, and these should be examined in basic studies and screening efforts, Borchelt said. Many different reasons may produce the disease process, and treatment strategies should be investigated accordingly.
New Models of ALS
Steps toward additional models for drug screening were presented at the workshop. Jiou Wang, Ph.D. at Yale is working with a worm that expresses mutant SOD1. Changes in the worm due to the mutation are easily measured and should serve as an efficient screen for candidate therapeutics.
Many proteins other than SOD1 are changed in other motor neuron diseases with some similarities to ALS. Shangxi Xiao, Ph.D., in the lab of Janice Robertson at the University of Toronto, is working on peripherin, a protein that plays a role in cellular transport and in nerve regeneration. The role of the Nogo proteins in transport and regeneration of nerve fibers is still unclear; with continued efforts of Noam Harel of the lab of Steven Strittmatter and Dupuis, this may be sorted out.
Andres Kottmann, Ph.D. at Columbia is investigating the function of a protein called wolframin in spinal cord motor neurons and in neurons of the fronto-temporal brain. Mutations in wolframin cause the hereditary, fatal neuro-degenerative disease, Wolfram syndrome. Mice deficient of wolframin have many similarities with ALS, including aspects related to the fronto-temporal dementia that affects some ALS patients. Wolframin appears to control the content of calcium ions in neurons and might be involved in the regulation of apoptosis, the form of cell death observed in ALS.
Martina Weidau-Pazos, M.D., Ph.D. at the University of California, Los Angeles presented a careful plan to apply the new science of proteomics to the ALS puzzle by comparing different models of motor neuron disease to see what differences might exist in the proteins expressed inside cells in those models. She will compare motor neuron damage inflicted by a mutation to the protein called tau, and an SOD1 mutation, to find any common set of proteins involved.
Genes that are active or resting can be probed by microarray, a technique to survey the status of genes in a cell. Weidau-Pazos and also Eva Hedlund, Ph.D. are taking this approach. Hedlund, who works with Ole Isacson, M.D., Ph.D.at McLean Hospital in Massachusetts, will look for active or less active genes in motor neurons that are relatively resistant to the effects of ALS, as compared to those motor neurons that are dying rapidly in the disease. Eran Perlson, Ph.D. is working with Erika Holzbaur at the University of Pennsylvania on proteins with a role in cell transport and regeneration using a proteomic approach as well to find any differences in ALS.
Stem Cell Avenues to ALS Therapeutics
Signals during development tell stem cells which types of cells to become and where to go. In the nervous system, these molecular signals are accordingly complex and must be orchestrated to allow successful stem cell therapy. Reports from Agnes Lukaszewicz, Ph.D. with David Anderson at California Institute of Technology and Paola Arlotta, Ph.D. at Massachusetts General Hospital in Boston gave progress in this area. Arlotta has helped uncover key signals in the birth of the neurons that originate in the brain and descend the spinal cord to control muscles. These cortico-spinal cells are among those that die in ALS. Better knowledge of the signals to generate these cells will help with design of effective stem cell therapies for nerve disorders such as ALS.
Fischbeck noted that progress in understanding the disease processes should be tuned to any possible therapeutic application, in order to best serve the patients and their families who have so little time. Robin Conwit, M.D., NINDS clinical trials coordinator, was impressed by the diversity of approaches to the challenge of ALS and stressed the need for rapid translation into clinical trials in the disease. Both explained funding opportunities available through the National Institutes of Health that could support more rapid clinical progress through a better understanding of the biology of the disease.
Workshop participants agreed that the meeting was a successful forum in which to be challenged by what others are finding and how to combine the different approaches toward successful therapy. One participant was impressed that scientists of such different expertise could cross disciplines to appreciate, integrate, and plan future approaches with the new knowledge imparted at the workshop. It is precisely this goal that ALSA seeks and has succeeded to implement. Future meetings will undoubtedly showcase the fruits of the collective effort.
