
Related recent news releases
First Fruits of Genome Wide Search Published for Sporadic ALS
Gene Screening On Track For Typical ALS
Newly Revealed Chromosome Locations Linked to ALS
AAN News: Genetic Findings Indicate Promising Therapies
Scientists announce two new genetic sites linked to familial ALS
Overview Summary
ALS can be inherited. But in 90 percent of cases it appears in people with no family history of the disease. The first gene to be found that is linked to ALS is that coding for an enzyme, copper-zinc superoxide dismutase (SOD1). Known mutations in SOD1 account for about 2 percent of all cases of ALS. Researchers are still working hard to solve how this gene change actually makes motor neurons die. Scientists funded by ALSA are among those narrowing down the hunt for other mutations that produce the disease. Investigators already have made great strides towards the details of the known SOD1 mutations, thanks to several lab models (see also laboratory models of ALS) that duplicate in laboratory animals what happens in the inherited disease. What are the Genetics of ALS? As in Alzheimer’s and Parkinson’s diseases, ALS can be inherited, but usually it appears in people with no family history of the disease. With inherited diseases, the cause is a change in the genes, called a mutation. The change in the DNA is reflected in a mistake in making a protein. Either the protein is not formed properly, or it is not made at all if the instructions are cut short by the genetic change.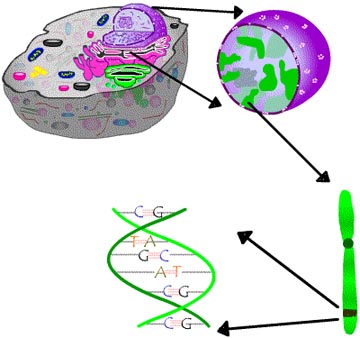 |
| The nucleus of a cell contains chromosomes, on which are the genes consisting of the DNA that codes for proteins, the main ingredients of living beings. |
In some cases of ALS, known mutations in the SOD1 enzyme make a change in this protein. In another mutation, linked to a very rare, juvenile form of ALS, a protein called alsin is no longer made.
For details on the genetics of ALS, click here.
In some diseases researchers have figured out exactly how mutation produces a problem. Often, despite knowing a mutation, scientists still lack a clear explanation for the resulting disease. For ALS, although the mutation is known for some inherited cases, researchers still are working hard to solve how the mutation makes motor neurons and their surrounding cells change function and die.
-
Sporadic ALS- the most common form of ALS- 90 to 95% of all cases.
- Familial ALS- occurring more than once in a family and accounts for 5 to 10% of all cases.
Only ten percent of ALS is inherited (also called familial ALS or FALS). Of these cases, a mutation is known for only about 20 percent. This means only about 2% of all patients with ALS have the SOD1 genetic change.
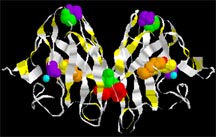 |
| The enzyme, copper-zinc superoxide dismutase {Cu/Zn SOD1}. The protein backbone of the molecule is indicated by the white ribbon. The molecules crucial to keeping the molecule in its proper shape are shown as colored balls: these include the copper and zinc ions that help the enzyme do its job of deactivating reactive oxygen. |
In 1991 a team of ALSA-funded researchers linked familial ALS to chromosome 21. In 1993 the research team identified the precise defect, a change in the DNA for the protein called copper-zinc superoxide dismutase 1 (SOD1). Researchers since have found more than 100 mutations in different places in the coded DNA instructions for making SOD1.
This protein is an enzyme that normally protects cells against damaging byproducts of cellular processes. But in ALS, the defective SOD1 does not appear to produce the disease by losing this protective function. Instead, evidence points to a new, toxic property acquired by the changed enzyme. Scientists are still seeking to find the molecular details of this acquired toxicity (see also SOD1 and ALS).
Additional Gene Possibilities Researchers are trying to pinpoint the exact genes responsible for other inherited cases of ALS. Finding different families with the disease in several generations has led scientists to probe locations on different chromosomes where genetic change may reside. These suspect regions are on various chromosomes, including those numbered 16, 18, and 20 (humans have 23 chromosomes referred to by numbers). Similar Clinical Scenarios ALS is remarkably similar whether it is inherited or appears in a person spontaneously with no family history of the disease. That is why researchers are so encouraged by knowing the mutations in SOD1. Therapy designed to address the consequences of the change in this protein may lead to treatment for all cases of ALS.Scientists funded by The ALS Association (ALSA) are among those narrowing down the searches for other mutations that produce the disease. But researchers already have made great strides into the molecular details of SOD1 mutation and ALS: several animal models are available that duplicate in the lab what happens in the inherited disease. These animals are engineered to express the human, mutated SOD1 protein (see also Lab Models of ALS).
In 2001, scientists announced a mutation in a gene that codes for a protein they named alsin. This gene is altered in a familial form of ALS, juvenile ALS 2. This form of ALS appears in relatively young people. The protein product of the gene is found in all cell types, including motor neurons. However the normal job of alsin is still a mystery. The mutation results in loss of its normal function. ALSA funded scientists are seeking to create animal models that will reflect the gene change and allow discovery of alsin’s normal role, and how it produces disease when changed.
In 2004 researchers funded in part by ALSA reported that a mutation in a gene on chromosome 9 coding for a protein called senataxin is responsible for a rare form of juvenile ALS. The exact function of senataxin is unknown.
